- 31 August 2016
- Posted by: nemcatgroup
- Category: Publications
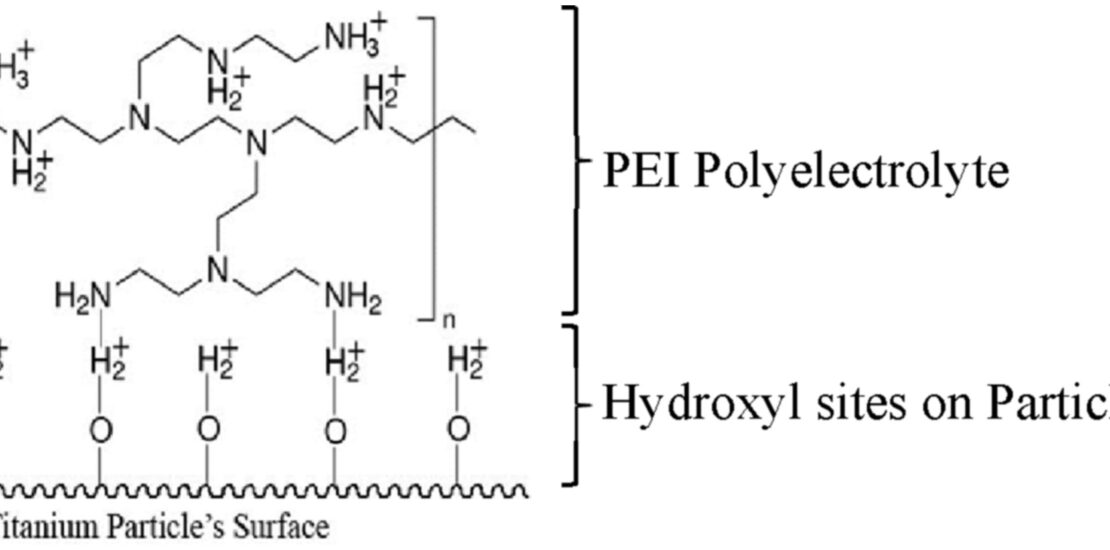
Proton activities of suspensions of Ti particles with added cationic polyelectrolyte as a function of acid additions have been investigated and compared in terms of the electrophoretic mobility and deposition yield. The proton activity in ethanol medium decreased with the addition of PEI polyelectrolyte and reduced further in the presence of Ti particles. The decrease in proton activity in the suspension indicates that protonation occurred on both the PEI molecules and Ti particles. It is proposed that the protonation of the amine groups of PEI and hydroxyl sites of Ti particle led to the formation of hydrogen bonding between the Ti particle and PEI molecules. Increase in the PEI and Ti with increasing acid addition translated to higher electrophoretic mobilities and deposition yield at low ranges of acetic acid addition (<0.75 vol%).
