- 23 February 2026
- Posted by: nemcatgroup
- Category: Publications
No Comments
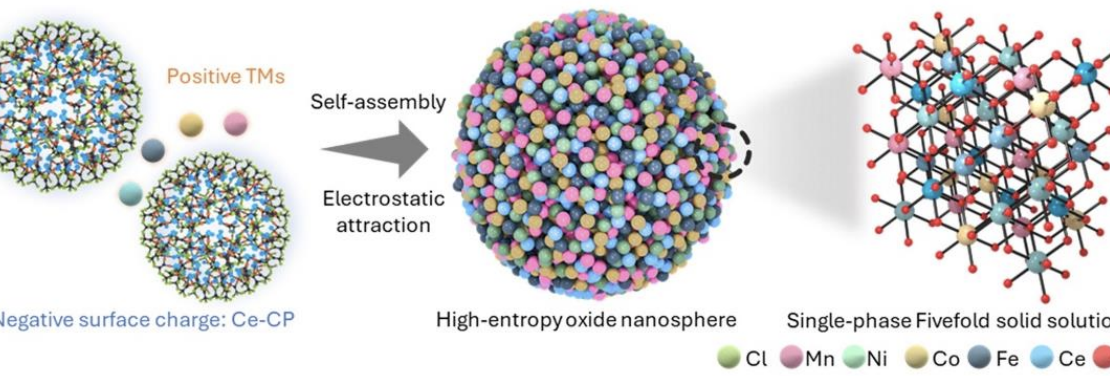
Research on high-entropy oxides generally is limited to elemental and structural interpretations applied to the performance of cathodes. However, there are only limited data on the principles of increasing disorder in terms of structural, electronic, and atomic mechanisms as materials convert from ordered to disordered. Zn-air battery cathodes are limited by slow kinetics, imbalanced oxygen evolution reaction charging, imbalanced oxygen reduction reaction discharging, and scalability (through the necessity of benchmark noble metals). The present work pioneers the engineering of multilevel disorder in high-entropy oxides, thereby transforming an intrinsically inactive matrix into a highly active cathode. Systematic modification of the disorder through increasing number of cations leads to the abrupt development of structural (2D defects), electronic (semimetallic conductivity), and atomic (low-coordination Ce) disorder. This multilevel disorder engineering of high-entropy oxides results in MnNiCoFe-CeO2 catalysts with stable active sites, rapid and balanced bifunctional (oxygen evolution/reduction reaction) performance, thereby promising Zn-air battery efficiencies and electrochemical durabilities greater than those of the benchmark materials).
